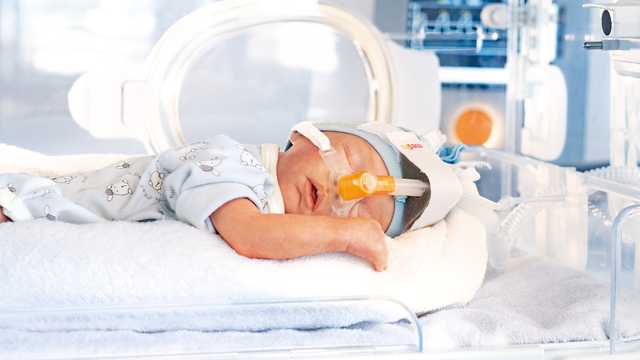
Apnoe und Atemunregelmäßigkeiten kommen bei fast allen Früh- und Neugeborenen vor. Je frühzeitiger das Kind geboren wurde, desto ausgeprägter tritt die Apnoe auf. Hierbei kommt es zu einer Unterbrechung der Atemmechanik und der Atemkontrolle. Das gravierendste Merkmal der Apnoe ist der nicht kontinuierliche Luftstrom in den Atemwegen des Patienten.
Apnoe ist für den Patienten aufgrund der Begleitsymptome wie Bradykardie <80 BPM (verringerte Herzfrequenz) und Hypoxie <80 % (verringerte Sauerstoffsättigung) klinisch relevant. Ziel der Prophylaxe und Behandlung von Apnoe ist es, negative Auswirkungen zu vermeiden oder zu minimieren (
Zusätzliche Atemhübe stimulieren die Atmung
Die Atemwege werden stabilisiert, wodurch die Atmung erleichtert wird
Die Eigenatmung des Patienten wird erkannt und das Gerät wechselt in den CPAP-Modus
Verfügbar bei den Geräten medinCNO, medinCNOmini, medin-NC3
Die Wirksamkeit der nasalen CPAP ist in diesem Kontext durch Studien belegt und Bestandteil der anwendbaren Richtlinien für die Behandlung der Apnoe und ihrer Begleitsymptome (
Die Wirkungsmechanismen sind:
Der medin ApnoeCPAP-Modus mit Apnoe-Behandlung kann von der Prophylaxe bis zur Behandlung einer bestehenden Apnoe eingesetzt werden.
Basierend auf dem nCPAP-Modus erkennt die einzigartige MediTRIG-Technologie zentrale und obstruktive Apnoen und reagiert mit automatischen zusätzlichen Atemhüben durch das CPAP-Gerät darauf. Die Höhe des Spitzendrucks der Atemhübe wird mit einem zweiten elektronischen Flowmeter eingestellt und zum Basisflow addiert. Sowohl die Parameter der ApnoeCPAP-Funktion als auch die des MediTRIG können individuell an den Patienten angepasst werden.
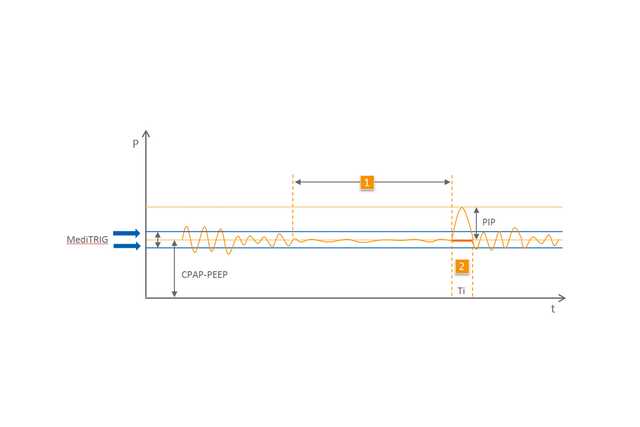
1 Apnoe
2 Automatischer Atemhub