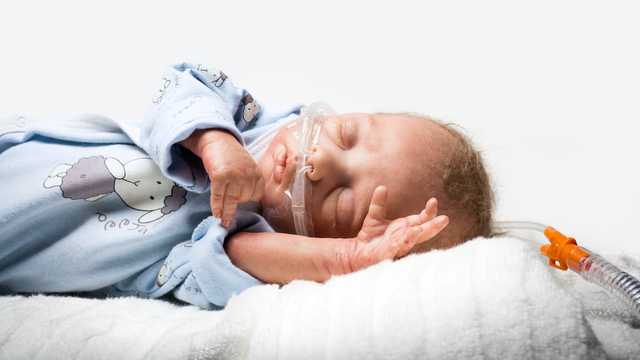
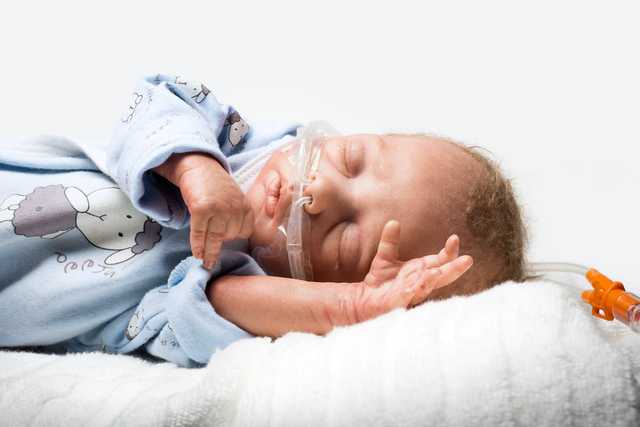
Nasal high-flow therapy (nHFT) describes the supply of heated and humidified breathing gas via a nasal cannula. The gas flow administered is thus higher than the patient’s inspiratory respiratory flow. In neonatology, a flow of more than 1 LPM is considered to be high-flow therapy, while for adults, correspondingly higher values should be applied. (
Other names for nHFT: High-Flow Nasal Cannula (HFNC) or Heated Humidified High-Flow Nasal Cannula. All of them designate the same form of therapy.
Easy and gentle application
Perfect complement to nCPAP respiratory support
Reduction in breathing effort
Available in the medinCNOmini, medin-NC3 and medinSINDI devices
If the breathing gas flows exceed the patient’s inspiratory flows, the supply of room air via the nose can be reduced or prevented, in contrast to the conventional low-flow nasal cannula therapy. As a result, the oxygenation and efficacy of the oxygen supply can be improved. (
The anatomic dead space is washed out through the breathing gas flow and thus reduced, which in turn can lead to a decrease in breathing effort. The carbon dioxide remaining in the upper airway at the end of expiration can also be washed out by the high breathing gas flow. (
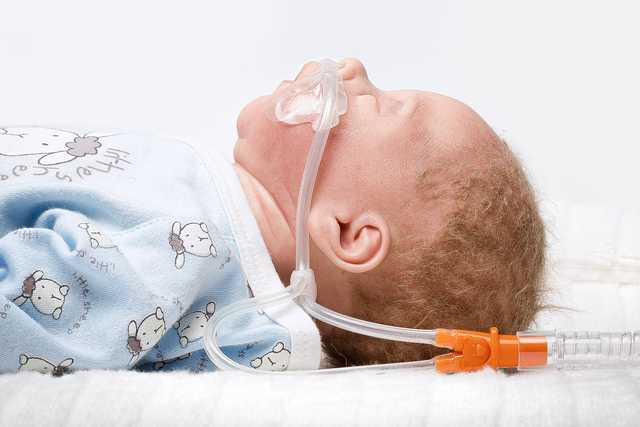
The application takes place in each case via our medinNuflow nasal cannulas and is thus very easy and gentle to the patient. To ensure the rinsing effect, the nasal cannula must be selected such that at least 50% of the nostril remains unobstructed, as intended leakage. The breathing gas flow can escape from the nose via this leakage.
No patient and tube pressures can be measured during nHFT. To protect the patient from undesired high pressures, it is necessary to maintain this leakage as a sort of overpressure valve. (
medin offers the nHFT application in the medinSINDI, medinCNOmini and medin-NC3 devices.
They can intuitively and easily switch from the nCPAP modes to the nHFT mode without the need to change the tubing system. The Medijet CPAP generator only needs to switch to the medin nasal cannula.
CPAP pauses, skin-to-skin care as well as treatment changes can be implemented easily and quickly. Your patients can receive flexible, individualized care as a result.
The following parameters are to be set